Dear visitors,
due to the current Corona situation, our student lab will be closed for the rest of the school year 2020/-21! New appointments will only be made again at the beginning of the new school year 2021/-22 (from September 2021)!
We ask for your understanding!
The team of the working group Didactics of Chemistry Jena wishes all students, parents and
teachers best health!
We are pleased about your visit on the homepage of our student laboratory and your interest in our offer!
Please note the following general information:
- Please discuss your visit to the student lab as specifically as possible with us in advance.
- The students are supervised by student assistants (chemistry student teachers).
- All materials (chemicals, run sheets, etc.) are provided, there is no cost to you!
- Upon request, we will also provide you with the material of the offered learning circles in advance.
- The experiments/content of the chosen topic should be prepared and followed up in class.
- Accompaniment by an expert teacher is expected.
Below you will find insights into the life of our student lab as well as a subsequent overview of the experiment topics offered. These are sorted by age group and each contains important information, such as a description of the topic, recommended prior knowledge or possible applications in science lessons.
Please do not hesitate to contact us if you are interested or have any questions!
We are looking forward to your visit in our student lab!
Your team of the working group Didactics of Chemistry Jena
We would like to point out that the offer of our student laboratory is neither intended nor desired for the organization of hiking days or other leisure activities, but serves to supplement science lessons.
-
Bionics - Learning from Nature
The dream of flying - which child has not dreamed it? The famous Italian artist, philosopher and engineer Leonardo da Vinci (1452 - 1519) also dreamed this dream and constructed a flying machine based on the model of bird flight. After the technical progress of the last centuries the dream came true and also man has conquered the sky today and flies, e.g. with airplanes, quite naturally through the air. However, this is just one of many examples of how role models from nature influence our lives. Think of water-repellent coatings, the salt shaker or the Velcro fastener. Making models from nature technically usable is the core idea of the still very young discipline of bionics. Based on the experiences with the learning set from the MNT and NwuT lessons, the practical course Bionics - Learning from Nature can also be found in the canon of student laboratory offers for elementary schools. The main focus is to show children the role of the natural sciences in our everyday lives using concrete examples. The set primarily uses playful approaches and pays particular attention to the fun of exploring bionic examples. The aim is also to train scientific observation and practical work (psychomotor skills) so that these can be further developed in later science lessons.
Recommended prior knowledge:
- no previous knowledge necessary
Tarentola gomerensis Joger & Bischoff, 1983, the "Gomera gecko" (taken in Valle Gran Rey, La Gomera).
Image: Marcel Simon -
Fragrances & Scents
When absorbing information from the environment, vision is undoubtedly the most important sense for humans. But hearing, tasting, feeling and especially smelling should not be underestimated! Because a smell can unfold also with humans unexpected effects: We associate specific situations in the course of our lives with certain smells. Scents also determine whether we feel attracted to another person or whether we tend to distance ourselves from them, true to the saying "I can't smell you!". And last but not least, many a fragrance literally makes our mouths water. In other animals and plants, however, odors play a fundamental role, determining friend or foe, evolutionary success or extinction. The chemical language of scents is considered the oldest form of communication in nature. The Fragrances & Scents course takes children on a journey of discovery into the realm of our sense of smell. From scent extraction and the production of their own scent stickers to the question of How quickly our nose actually gets used to smells?, this topic is explored in a playful, experimental way.
Recommended prior knowledge:
- no previous knowledge necessary
Flower of Tripleurospermum maritimum agg.
Image: Marcel Simon -
Sensory Perception
With great interest and insatiable curiosity, man has always dealt with processes in the natural world around him. In these processes of scientific work, human sensory perception and objective observation play an essential role. The senses enable even young children to explore natural phenomena and thus their own environment by looking, touching, smelling, hearing and tasting. In this context, the Sensory Perception course is not only suitable for MNT lessons, but also for gaining initial scientific experience in preschool and elementary school. In this course, experiments are conducted that are visually and tactilely appealing and are intended to encourage children to ask questions about nature. The children are thus introduced to the process of scientific work in an age-appropriate manner. In addition, the practical course also addresses the promotion of key scientific skills, e.g. describing, measuring or drawing conclusions and, last but not least, the development of psychomotor skills.
Recommended prior knowledge:
- no previous knowledge necessary
Hearts (taken at the Museum of Illusions, Vienna)
Image: Marcel Simon -
Water-Soil-Air
Based on the experience gained in MNT lessons, the Water-Soil-Air learning circle was also fundamentally revised as an offering for elementary schools, especially with regard to the subject areas 'water' and 'air', and expanded to include several experiments. The aim is to enable children to learn about the properties of these substances and to classify them, for example, according to their states of aggregation or their appearance (solids, liquids, gases). With the help of effective and age-appropriate experiments, experience is gained with scientific laws, e.g. density of substances: Which materials float, which sink in water? or material properties are actively acquired by the students. On the one hand, the learning circle conveys the underlying technical content in a child-friendly way and, on the other hand, introduces basic concepts of science and scientific work in a playful way: The children are consciously encouraged to form hypotheses/ask questions, are encouraged to answer them experimentally and are given the freedom to consciously vary the experimental conditions.
Recommended prior knowledge:
- no previous knowledge necessary
Water, Soil and Air (taken at Loch Tay, Scotland)
Image: Marcel Simon -
Wonderful Colors
'Colors are the smile of nature [...]' Leigh Hunt, 1784 - 1859. Be it a rainbow, a sea of colorful flowers or the colorful splendor with which the deciduous trees clothe themselves in autumn: Colors are among the most fascinating phenomena in the world around us. It is therefore hardly surprising that the topic of "colors" is present in so many areas of our society, whether in art, literature, the natural sciences or even philosophy. Colors are ascribed a wide variety of meanings and emotions: 'red with anger' or 'yellow with envy'. Children in particular are fascinated by colorful natural phenomena and ask questions about nature: How do colors come into being? or Why do things have these colors in particular? The Wonderful Colors course aims to get to the bottom of some of these children's questions and create a natural science-discovery approach to this topic. In this practical course, the students can make different colors themselves with natural materials, extract leaf dyes, examine them, dye textiles with plant dyes, or even discover the phenomenon of fluorescence.
Recommended prior knowledge:
- no previous knowledge necessary
Autumn colors (taken in Wurzbach, Thuringia)
Image: Marcel Simon
-
Bionics - Learning from Nature
Man has always been observing nature, taking inspiration from it and trying to imitate it in its form and mode of action. One of the most famous examples of this are Leonardo da Vinci's drawings for the construction of flying machines, which are based on the model of bird flight. It was not until 1960 that bionics was born as an independent discipline. Bionics is a portmanteau of the terms biology and technics. Today we find a variety of everyday objects or products that are based on models from nature, such as the salt shaker. In the learning set Bionics - Learning from Nature, students are introduced to the topics of this still young, interdisciplinary science. By working through a total of eleven stations, including, for example, the lotus effect, folding in nature, the Velcro fastener or the honeycomb, the students are given an insight into the nature of bionics and the direct connection between natural models and everyday products is demonstrated.
Recommended prior knowledge:
- no previous knowledge necessary
Immature capsule of Papaver somniferum L.
Image: Marcel Simon -
MNT I - Sensory Perception
Beyond all subject boundaries of the natural sciences, the conscious handling of human sensory perception as well as targeted and objective observation form one of the necessities and foundations of scientific work. In the Sensory Perception course, students work on experiments that are visually and tactilely appealing as well as designed to encourage proper research. Students are gradually introduced to the nature of scientific work. Thus, the focus of this learning circle, in addition to the training of the skills of observation, is also the development of the key skills of describing, measuring, reasoning and the training of psychomotor skills. Thus, this learning circle is suitable for the integration into the science lessons in the subject Man-Nature-Technology (MNT).
Recommended prior knowledge:
- no previous knowledge necessary
Curved squares (taken at the Museum of Illusions, Vienna)
Image: Marcel Simon -
MNT II - Structure of Substances
'That I may know what holds the world // together at its innermost core.' This famous quote from Johann Wolfgang Goethe's Faust. The Tragedy of the First Part illustrates, more than any other, man's quest for knowledge about the structure of matter. Young students also bring a wide variety of ideas about this topic with them to their early science classes. For this, especially for the subject MNT, the practical course Structure of Substances was developed. Similar to the learning unit on the topic of 'sensory perception', this learning circle also addresses scientific working methods such as observation, investigation and, above all, experimentation. The associated practical activities are also intended to introduce the students to independent research-based learning in the natural sciences. However, the focus of this learning circle is on the concept of matter and its abstraction to the particle level, with the help of age-appropriate models. Thus, topics such as pure substances vs. mixtures of substances and substance separation are addressed in the experiments.
Recommended prior knowledge:
- terms 'body' and 'substance'
- ideas about the particle model (spherical particle model)
Models for the structure of substances
Image: Marcel Simon -
MNT III - Water-Soil-Air
Water, soil, air and fire - all earthly things are made up of these elements in certain proportions, according to the Greek philosophers' concept of the four elements. This theory has its roots in the works of Thales of Miletus (624-546 B.C.) and retained a central position in chemistry until the 17th century. Even today, the idea of these 'basic elements' can be found in esotericism, art, as well as comics and movies, as in the Fantastic Four from the Marvel comics. The Water-Soil-Air course allows students to explore three of these 'elements' from a scientific perspective using effective and age-appropriate experiments. The focus is on making the scientific phenomena and material properties tangible. At the stations, typical student ideas can be worked on, which can lead to learning obstacles for students in dealing with the particle model in the subject areas of water, soil and air.
Recommended prior knowledge:
- substance term
- ideas about the particle model
Water, Soil and Air (taken at Kingussie, Scotland)
Image: Marcel Simon -
MNT IV - Food & Nutrition
'Eating and drinking whole foods keeps you healthy and promotes performance and well-being.' - German Nutrition Society. Today, it is a general consensus that a balanced diet plays a central role in maintaining the health of the human body. Supermarket shelves offer consumers a veritable plethora of different foods: Which ones are beneficial to health? What should be eaten in moderation? Recommendations are provided by overviews such as the nutritional circle or the well-known food pyramid. In order to eat healthily, however, a sound knowledge of the ingredients of the products we consume is essential! In the course Food & Nutrition, the students experiment with quite common everyday products and experimentally prove, among other things, the content components. The main focus is on the 'three major nutrient classes' (proteins, carbohydrates and fats) and vitamins.
Recommended prior knowledge:
- substance term
- ideas about the particle model
Healthy diet
Image: Marcel Simon
-
Chemistry in the Pencil Case
The oldest known drawings are Stone Age finger carvings in the Pech Merle cave near Cabrerets in France and date from a time around 23,000 BC. They were carved into the thin clay layer of a cave ceiling. The first 'writing instruments' were the fingers. In addition, wood, stones, tufts of grass and lumps of paint were used. In the course of time a variety of different writing utensils developed from this origin. Today, despite the advance of digitalization, the pencil case is still part of the 'basic equipment' of every student at school. A look inside shows the diversity of writing and other makeshift tools available today. From ink, the metal sharpener and erasers to a plethora of different pens: pencils, ballpoint pens, felt-tip and colored pencils, ink erasers or highlighters. The Chemistry in the Pencil Case lab course for schoolchildren focuses on these everyday objects and shows experimental approaches to examining them from a chemical point of view. The aim is to establish a direct link between the students' everyday lives and the subject of chemistry. The experiments in the course focus on ink, dyes, metal sharpeners and pencil leads and address the chemical topics of chemical reaction, redox chemistry, substances/substance properties and combustion.
Recommended prior knowledge:
- substance concept and substance properties (solubility, electrical conductivity, etc.)
- characteristics of chemical reactions
- simple redox term (definition via oxygen)
- Handling of the Bunsen burner
Separation of substances by paper chromatography
Image: Marcel Simon -
Glass
When people talk about a 'glass world' today, they are not referring to glass as a material per se, but rather to one of its most important properties, transparency. Glass is one of the oldest and chemically most interesting materials of all, and we encounter it in many different forms: As window glass, in the form of drinking glasses, on mirrors or as the good crystal glass. The oldest glass finds come from Egypt and eastern Mesopotamia and are dated to the 3rd millennium BC. The first small glass vessels appeared in Egypt as early as 1500 BC. The coloring by admixture of metal oxides was already known at that time. For some time now, the city of Jena has also been known for its glass tradition: Otto Schott made a name for himself here as early as 1875 with the manufacture of special optical glass. Schott, Abbe and Zeiss built up the production of optical glass in Jena in the following years. Glass is also interesting for chemistry lessons: from a chemical point of view, glasses are understood to be polymer solids that do not consist of a basic lattice that is regularly repeated, like a crystal, but rather are built up from a network without rules. They are therefore also referred to as 'frozen liquids'. In the Glass lab, students can experimentally investigate the resulting properties and produce colored glass and an enamel coating themselves.
Recommended prior knowledge:
- substance concept and substance properties
- safe handling of the Bunsen burner
Glass
Image: Marcel Simon -
Household Cleaners - Acid-Base Chemistry made easy!
In every household, washing, rinsing and cleaning is done almost every day. As consumers, we now have a wide range of products to choose from. Behind this is a huge industry: In Germany alone, a total of around 1.3 million tons of cleaning agents and detergents are sold to private consumers every year. This figure does not yet include commercial and industrial cleaning agents! Broken down more specifically, this amounts to 630,000 metric tons of detergent, 220,000 metric tons of fabric softener and 480,000 metric tons of cleaning and care products per year. (Source: Federal Environment Agency of GermanyExternal link)
In 2020, the detergent industry generated sales of 1.2 billion euros with household cleaners alone. If detergents, rinse aids, polishes, room fragrances and other products are also included, sales are as high as 4.6 billion euros. (Source: StatistaExternal link)
Many cleaning agents are optimized for special applications and contain reactive substances. The students are already familiar with many of these products from their everyday lives. In the practical course 'Household Cleaners - Acid-Base Chemistry made easy', the main focus is on acidic and basic household cleaners and the associated chemical reactions. The students can thus acquire and consolidate important basics of acid-base chemistry by means of everyday-oriented experiments.Recommended prior knowledge:
- acid-base definition according to Arrhenius
- structure and dissociation of acids and bases (on particle level, ion concept)
- terms pH value, indicator and neutralization
download experiment scriptpdf, 4 mb · de
The 'red cabbage color series'
Image: Marcel Simon
-
Bionics - Learning from Nature
Leonardo da Vinci, who wanted to realize the dream of flying, formulated at the beginning of the 16th century: 'The human creative spirit can make various inventions [...], but it will never succeed in one that would be more beautiful, more economical and more straightforward than that of nature, because in her inventions nothing is missing and nothing is too much.' Man would like to take advantage of these various biological inventions. A separate field of research has existed since the mid-1960s that looks at nature's patterns and attempts to transfer them to technology, called bionics (a portmanteau of biology and technics). In this internship, the topics of this still young, interdisciplinary science are worked on, which has already produced a multitude of marketable products that are an integral part of our everyday life. By working through a total of eleven stations in the practical course, for example on the lotus effect, folding in nature, the Velcro fastener or the honeycomb, the students gain an insight into the nature of bionics. After consultation, this practical course can be extended to include the topics of construction bionics (functionalized fibers, stability gain through the method of tension triangles) and process bionics (Grätzel cell).
Recommended prior knowledge:
- no previous knowledge necessary
Immature capsule of Papaver somniferum L.
Image: Marcel Simon -
Detergent - repackaged!
'Soap is a measure of the prosperity and culture of states. It is certain that when comparing two states with the same number of inhabitants, one can declare the richer, more prosperous and more cultured one which consumes the most soap.'
(Justus von Liebig, 1803 - 1873)Even if, in reference to this quotation from Justus von Liebig, soap is attributed a special role with regard to detergents, water should not be underestimated as a basic detergent. However, as a polar solvent, it cannot dissolve non-polar substances such as fats from our skin or textile fabrics. This is why people throughout history set out in search of a better formulation: from the Sumerians, the Egyptians and Romans, through the Middle Ages to industrialization and modern times, soaps as well as detergents developed to their current abundance. This topic also has a long tradition in chemistry classes. And so, in the practical course Detergents - repackaged!, numerous classes of substances such as surfactants, organic dyes, builders, enzymes as well as simple inorganic salts are examined in an everyday-oriented manner. Easy-to-perform detection reactions, historical aspects, the production of own soaps as well as impressive experiments on surface tension and the Tyndall effect are the focus. Furthermore, ecological consequences and sustainable development are to stimulate discussion and contribute to the development of the students' interests.
Recommended prior knowledge:
- knowledge of central organic substance classes and their functional groups (especially alcohols, carboxylic acids and esters) as well as saponification reactions.
- confident handling of organic structural formulas
- derivation of substance properties from their structure (structure-property relationship)
- structure, divisions and micelle formation of surfactant particles
- term indicator (knowledge of the indicator phenolphthalein is ideal)
Detergent
Image: Marcel Simon -
Drugs & Pharmaceuticals
The principle of using organic compounds against infections has been known since ancient times. An important milestone in recent history was reached in 1929: Alexander Fleming (1881 - 1955) discovered penicillin with its antimicrobial effect primarily against staphylococci and streptococci, two bacteria that cause a large number of human infectious diseases. It counts as the first antibiotic. In 1945, Fleming, along with Howard Florey and Ernst Boris Chain, received the Nobel Prize in Medicine for it. Since then, the consumption of medicines has also been subject to steady growth in Germany: for example, the consumption of finished medicines in 2012 amounted to almost 40 billion prescribed daily doses. In a European comparison, Germany is one of the countries with the highest expenditure on pharmaceuticals per inhabitant. But pharmaceuticals play a role not only for medical applications. They are also part of current scientific research and are integrated into the pharmaceutical industry. The practical course on Drugs & Pharmaceuticals focuses on the areas of natural remedies, analgesics and antacids. Here, students conduct experiments with commercially available analgesics and gastric acid regulants. Examples include investigations into the dissolving behavior of enteric-coated tablets, the detection of vitamin C in combination preparations, the presentation and hydrolysis of ASA or the quantitative analysis of ibuprofen.
Recommended prior knowledge:
- knowledge of central organic substance classes as well as their functional groups (especially alcohols, carboxylic acids and esters)
- confident handling of organic structural formulas
- reactions of acids and bases (especially neutralization reactions)
- terms pH value and indicator
- concept of aromaticity
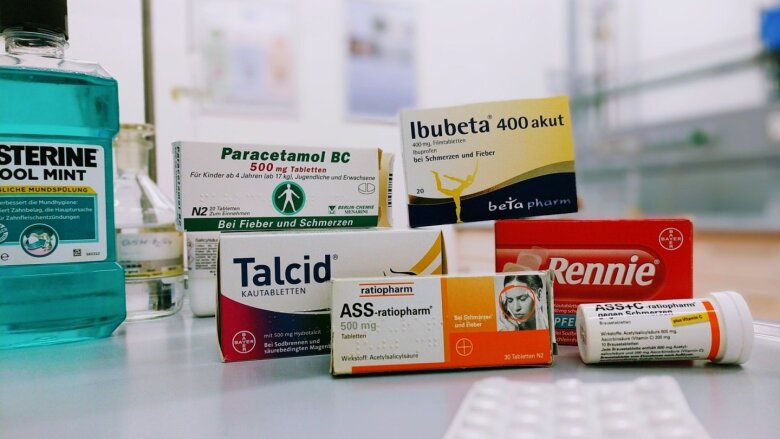
Various medicines
Image: Marcel Simon -
Fragrances & Flavorings
Scents play a central role in nature! Many animals emit pheromones. These are chemical messengers that trigger certain behavior in other animals or alter hormone secretion. Plants often produce scents in concentrated form as essential oils. The scents of plants serve primarily as attractants. While in humans, olfactory perception is often considered less important than sight and hearing, odors do have a direct, but often unconscious, influence on our emotional life and attitudes. In humans, body odors, including those from breath, skin secretions, breast milk, and sexual secretions, play a role in the social environment. We can store odors in our memory for a very long time, usually evaluating them as pleasant or unpleasant memories. In addition, they also play a decisive role in food intake, and today the food industry uses a whole range of natural and synthetic aromas. In the Fragrances & Flavorings practical course, students examine various scents, learn about different processes for obtaining scents, produce scents themselves and test their own olfactory perception. From a chemical point of view, the ester group of substances is particularly addressed.
Recommended prior knowledge:
- knowledge of central organic substance classes as well as their functional groups (especially alcohols, aldehydes, carboxylic acids and esters)
- confident handling of organic structural formulas
- derivation of substance properties from their structure (structure-property relationship)
- term indicator
Red roses
Image: Marcel Simon -
Regenerative Fuels
Increasing mobility of the population and growing demand for energy sources from emerging nations such as the People's Republic of China or India mean that more and more fuels are needed. On the other hand, there is only a finite supply of fossil fuels on our planet, the extraction of which is associated with increasing environmental risks. Due to this problem as well as ecological and climate policy goals, the European Union in particular is now committed to the use of regenerative energy sources. Consideration of the use of fuels based on alternative raw materials began much earlier. For example, as early as 1860, N. A. Otto used bioethanol ('potato spirit') as a knock-resistant fuel for the prototype of the internal combustion engine. Nevertheless, bioethanol failed to gain acceptance due to the better availability and low price of gasoline. Today, energy policy issues such as anthropogenic carbon dioxide emissions, regenerative fuels and their critical reflection are permanently present in the media, most recently through discussions about diesel driving bans or environmental badges. The practical course on Regenerative Fuels focuses on the topics of biodiesel and bioethanol. The students learn to compare these with conventional fuels from various points of view. They can even produce biodiesel or bioethanol themselves.
This course is only possible by special arrangement!
Recommended prior knowledge:
- knowledge of central organic substance classes as well as their functional groups (especially alcohols, carboxylic acids and esters)
- confident handling of organic structural formulas
- structural composition of carbohydrates, fats and oils
- alcoholic fermentation
- derivation of substance properties from their structure (structure-property relationship)
Flowering rape (Brassica napus L.)
Image: Marcel Simon -
Stimulants
The coffee as a pick-me-up in the morning - the number 1 popular drink of the Germans, even before the beer! Both belong to the so-called stimulants. These are foods that are consumed by people not because of their nutritional value, but because of their stimulating effect or taste. They have occupied an important position in all human cultures since time immemorial, both on an individual and social level. Globally widespread stimulants include alcohol, coffee, tea, cocoa and tobacco. Mate, kola, coca, betel, qat, opium, kawa kawa, etc. are of more regional importance. The active ingredients are mainly alkaloids, which have a stimulating effect on the cardiovascular system and the nervous system. The dose is decisive for the positive or negative effect. The boundaries between stimulant and intoxicant or addictive drug are often blurred. In addition to the active ingredients, stimulants contain other substances such as nutrients, vitamins, enzymes, colorants, flavorings and dietary fibers, which can be the subject of chemical studies. In the practical course on Stimulants, the focus is on the experimental investigation of coffee, tea, cocoa and chocolate with regard to their constituents. The focus is on typical detection methods, e.g. of starch, cellulose, proteins or tannins.
Recommended prior knowledge:
- confident handling of organic structural formulas
- structural composition of carbohydrates, proteins and fats
- detection of reducing sugars using the Fehling sample
- term indicator
A typical stimulant: coffee
Image: Marcel Simon -
Titration
The term Titration is generally understood to mean a quantitative, measurement-analytical method in which the determination of an unknown quantity of a dissolved substance is carried out by careful addition of a reagent solution (standard solution). The concentration of the measuring solution used is known. In this way, characterizations concerning the composition of a solution (analyte), e.g. content or concentration determinations, can be carried out. This plays a role in various areas of everyday life, for example in the determination of the content of certain ions in our drinking water. In order to determine the concentration of a dissolved substance, there are various ways of carrying out a titration. In the practical course of the same name, the students learn five titration methods. These include the 'classic' indicator-based acid-base titration on the one hand and more advanced forms such as potentiometry, conductometry, complexometry and iodometry on the other. These are explored experimentally in the context of everyday topics, such as the phosphoric acid content in cola, the total acid content in white wine or the determination of water hardness. Furthermore, the students compare titration curves of both mono- and multi-protonic as well as weak and strong acids with each other.
In consultation, this practical course can also be designed for targeted preparation for the Abitur with regard to potentiometry and conductometry on the basis of original Abitur examination tasks from recent years!
Recommended prior knowledge:
- chemical equilibrium (acid-base equilibria), law of mass action
- acid-base concept according to Brönsted (donor-acceptor principle)
- mathematical definition of pH and pOH values as well as strength of acids and bases
- neutralization, buffer and indicator (knowledge of "common" indicators is ideal).
- describe dimensional analysis as a quantitative method and important equipment
(e.g. burette)
Titration
Image: Marcel Simon
August-Bebel-Straße 2
07743 Jena
Opening hours:
by appointment
Contact person:
Marcel Simon